In September 1977, I had a very high fever. I was nine months old, so of course I don’t remember it, but my mother most certainly does. After all, what mother wouldn’t remember the heat that would not break, or the stillness that comes over a sick infant, all the while contending with a worry that has no name except itself. I never developed a rash, and soon enough the fever passed and I recovered, and whatever caused it went unrecorded outside of a simple “fever.” Nobody knew yet what the fever had done to my hearing.
My family started noticing something was off when I was around two or three, that I wasn’t responding the way a child should. There was a distance between me and sound that shouldn’t have been there. I wasn’t fitted with hearing aids until I was five. By then it was 1981, four years after the fever, and the cause listed in my record hadn’t changed. Still just “fever.” Still no explanation for what came after.
Nobody mentioned measles, since there had been no rash. That is entirely reasonable thinking because everyone associates a rash with measles and it is the most telling signature that makes a diagnosis legible. Without it, the fever was just a fever, the hearing loss that followed was just a hearing loss, and the two facts lived in separate compartments of my family’s understanding for decades before I started connecting them myself.
I study Health Information Management, which means I spend a lot of time thinking about medical records, about what gets written down, what gets coded, and what gets lost between those two things. A few years ago, when I needed measles titers for an internship, I discovered that I had no records of a past infection and no way to confirm whether I had received more than the single dose of MMR. The titers came back unusually elevated, the kind of result you see in an immune system that was primed by natural infection before vaccination and then mounted an amplified response on top of it. A fever without a rash in a nine-month-old in 1977 is consistent with atypical measles, which tends to occur when maternal antibodies partially suppress the immune response, blunting or even eliminating the rash altogether while the virus does its damage to structures in the inner ear.
Measles was circulating in the United States at tens of thousands of cases per year in 1977, well before the two-dose schedule brought the numbers down. This isn’t hard proof, but the pieces fit too well for me to ignore. I’ve started thinking of my hearing loss as probable measles, unconfirmed, never coded, and never linked. From what I’ve seen in my studies, that’s how a lot of these cases stay buried.
The rash didn’t appear, so nobody wrote it down; because nobody wrote it down, the idea of measles never traveled through time the way a coded diagnosis would have. I figured it out myself, nearly fifty years later, by going to the literature and the immunology and the epidemiology of 1977, and finding how they lined up. That process is, in miniature, exactly what this article is about.
Vaccination rates are dropping, so old diseases are resurfacing, thanks, in part, to too many people basing health choices on loud claims with zero backing. Unfortunately, most of those claims usually have a tiny kernel of truth, and that’s why they’re so hard to argue against. Data is the best counter we have. It’s not neutral, sure, but at least you can check where it came from and poke holes in it.
When you follow the numbers on measles and hearing loss, what you find is that the surveillance record has a structured silence built into it. Not intentionally, mind you. This isn’t something generated by bad faith, but made by a series of perfectly reasonable-sounding decisions about clinical coding that pile up over decades into something that makes certain kinds of knowledge essentially impossible to produce from existing records.
Before the measles vaccine became available in 1963, the United States reported between 319,000 and 763,000 measles cases per year. Those are the known and reported cases only. The CDC estimates the true infection count was closer to three or four million annually, which gives us a ratio of roughly six and a half true infections for every one that made it into surveillance data.6,7 Measles was so perfectly ordinary that most of it went undocumented because it was managed at home with only the most severe cases finding their way into a clinical record.
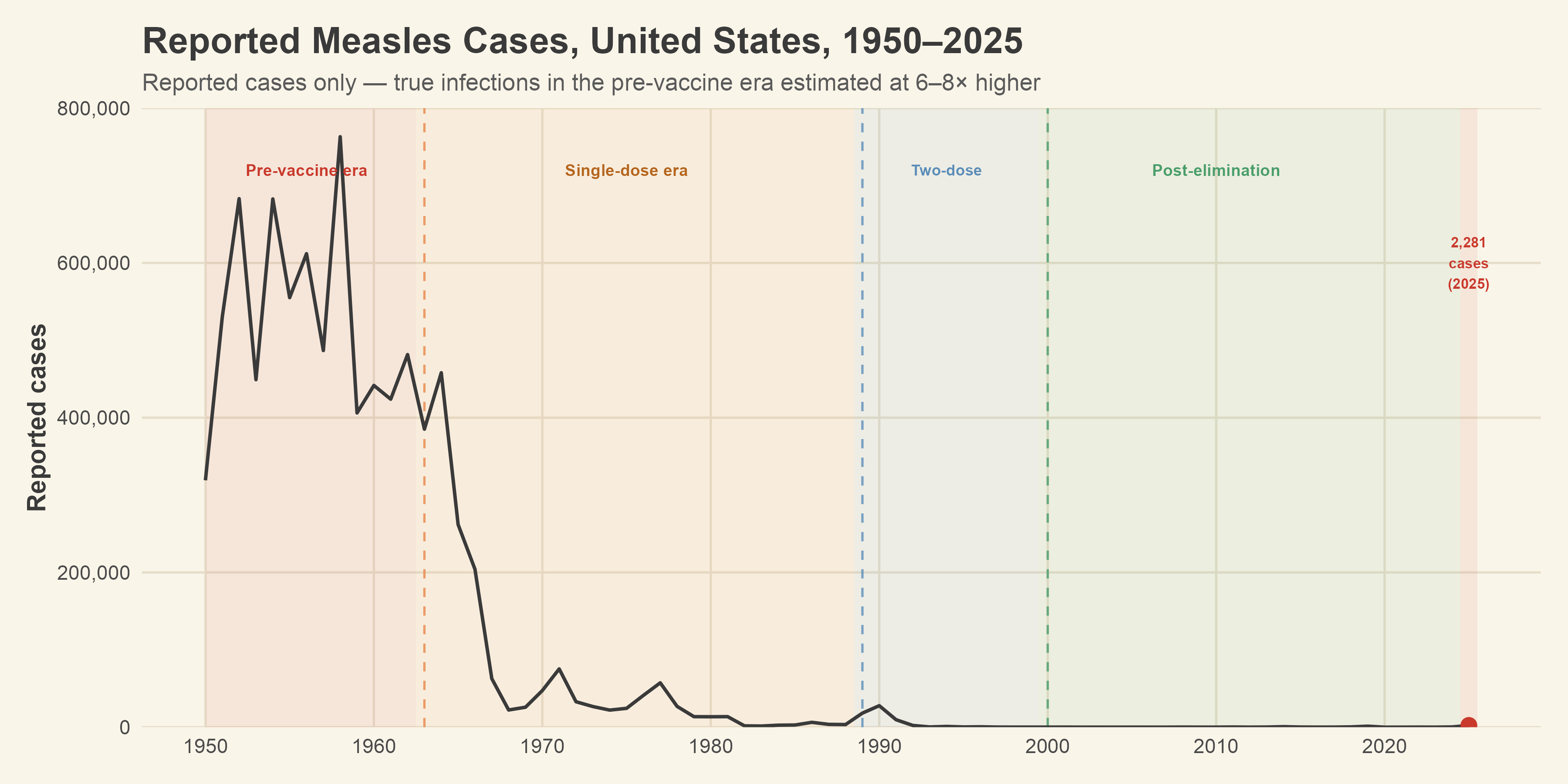
[Figure 1: Reported measles cases, United States, 1950–2025, with vaccine era annotations]
Cohen, Durstenfeld, and Roehm’s 2014 review in Trends in Hearing put the hearing loss complication rate at between 0.5% and 1.3% of measles infections.1 That range accounts for the otitis media route, where a middle ear infection leads to sensorineural damage through inflammatory toxins crossing the round window membrane, and the direct cochlear route, where the virus itself destroys hair cells and spiral ganglion neurons in the basal turn of the cochlea.1,14 When you apply that range to the true infection estimates for the pre-vaccine era, the projected annual hearing loss counts land somewhere between 25,000 and 45,000 new cases per year. Across the full fifty years from 1950 to 2000, the cumulative projected total reaches approximately 1.4 million cases of measles-associated hearing loss.
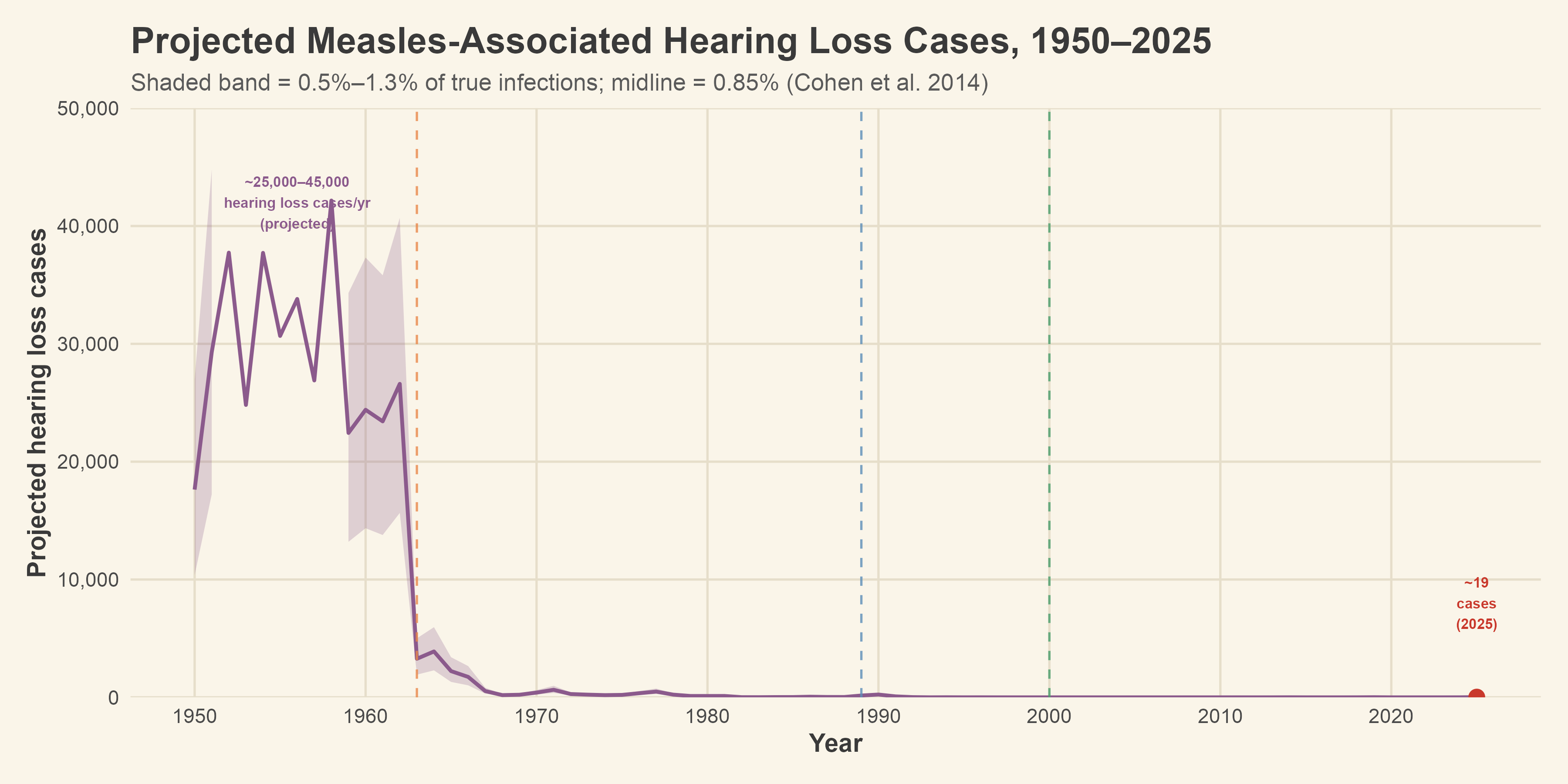
[Figure 2: Projected annual measles-associated hearing loss cases, 1950–2025, with uncertainty band]
None of those projected cases appear in measles surveillance data. Each one was coded as otitis media or sensorineural hearing loss, filed under H66 or H90, while the measles was documented separately under B05. The two were never connected, and then time passed and the information effectively disappeared into the ether. The biology is clear, the auditory literature spells it out, and people like me carry the titers that scream prior infection; but none of that shows up in surveillance data, and that’s exactly where it needs to be if we’re going to inform policy.
Here’s why this happens. U.S. medical coding is encounter-based, and the problem is baked into the structure, not some deliberate cover-up. Every clinical visit generates diagnosis codes that reflect what was assessed and treated at the time, and those codes get submitted for billing, then eventually aggregated into surveillance data. A sequela framework exists within ICD-10-CM, which allows a coder to attach the original cause to a downstream complication years later. Measles even has a specific sequela code, B05.3, for measles complicated by otitis media. The mechanism for doing the coding correctly is right there in the codebooks. What is not there, in most clinical settings, is any workflow that applies it reliably.
When a child develops hearing loss six months after a measles infection, for example, the audiology appointment happens at a different facility with a different provider. That provider’s awareness of the original measles encounter depends entirely on whether the information traveled with the patient through the system. The provider would need to know about the prior infection, think to connect it to the current presentation, and work in a system that supports retrospective linkage across providers. Almost none of those conditions are routinely met in a fragmented healthcare system. The hearing loss gets coded as H90, the measles goes unmentioned, and the connection dissolves. In 1977, nobody even had an electronic health record to not link to. The information simply never existed in any form that could travel.
The 2025 outbreak gave us a real-time case study in exactly how that dissolution happens and where it hits hardest.
The outbreak began in Gaines County, Texas, an agricultural community of roughly 22,000 people on the flat plains of West Texas, home to a substantial population of Low German Mennonites with historically low vaccination rates. By the time Texas DSHS issued its final report in August, Gaines County had recorded 393 confirmed measles cases. That is more than any other county in the country, more than most entire states, and more than the national total in any single post-elimination year before 2025.2,5,8 Transmission spread through linked chains into 23 other West Texas counties and eventually into New Mexico, Oklahoma, and Kansas, reaching 2,281 confirmed cases nationally by year’s end.2,5,8,9
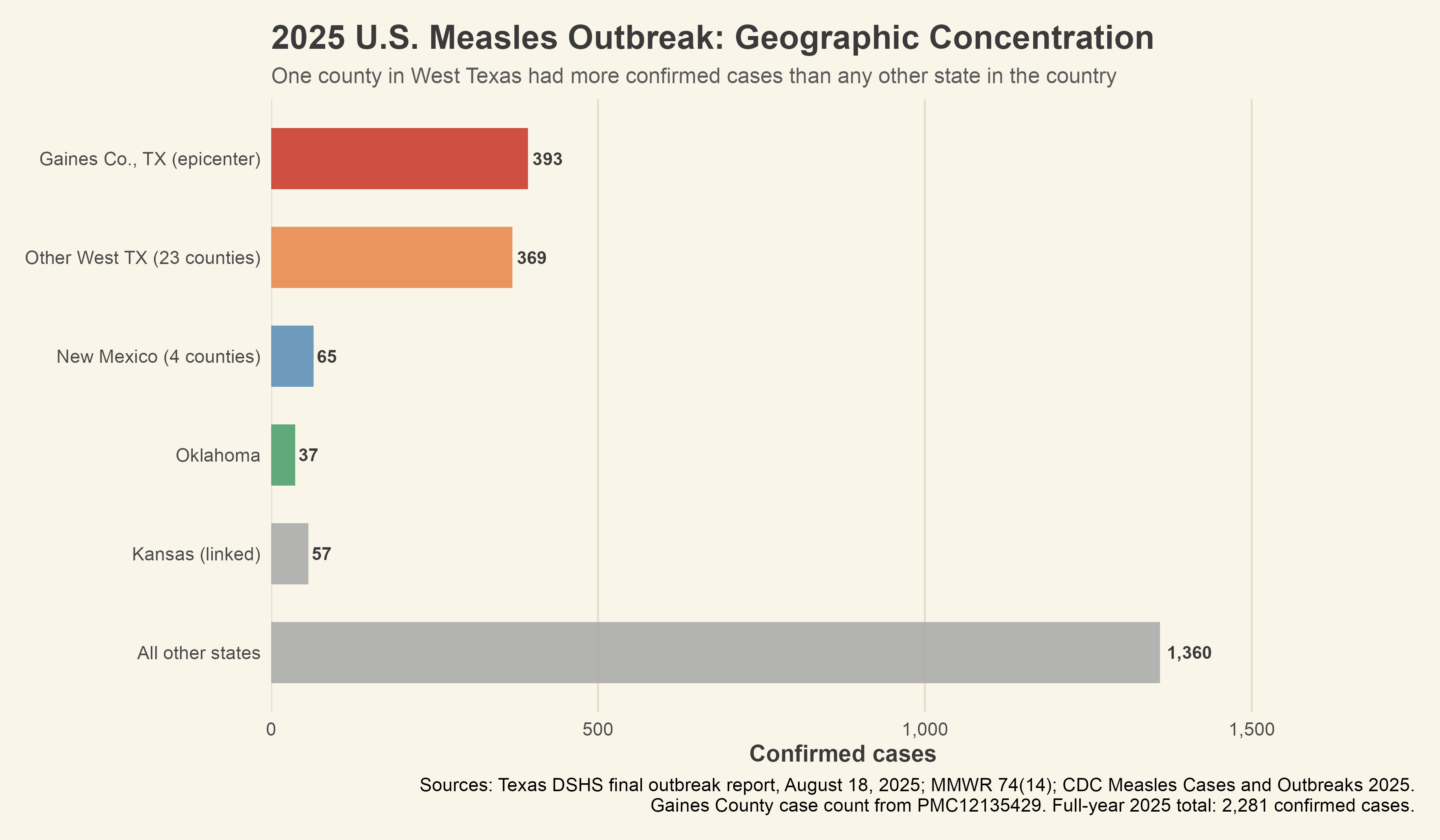
[Figure 3: Geographic concentration of 2025 outbreak cases by location]
Sixty-nine percent of those cases were children under 20, and 31% were under five. The age spread matters because young children are at the greatest risk for the long-term complications that follow otitis media in developing ears. To make matters worse, Gaines County has no audiologist on record in the NPPES provider database. The nearest audiology services are in Lubbock, about 60 miles north, or in Midland, about 90 miles southeast. For uninsured families, undocumented residents, and members of a community whose trust in mainstream medicine has long been low, the distance to care functions less as an inconvenience than as a brick wall. Garuccio and colleagues, writing in JAMA Otolaryngology in 2025, documented that 75% of U.S. counties qualify as hearing healthcare shortage areas, with rural counties disproportionately represented.3 Gaines County’s situation, while severe, is not entirely unique.
The surveillance funnel that results from this can be modeled per thousand measles cases, and the numbers are worth walking through because each step involves a loss that seems reasonable by itself but devastating in aggregate. Cohen et al.’s 0.5% to 1.3% hearing loss rate translates to between 5 and 13 children per thousand who will develop some degree of measles-associated hearing loss, whether through the otitis media pathway or direct cochlear damage.1 Using the midpoint of 0.85%, roughly 9 children per thousand will be affected. The question is how many of those nine will ever get a diagnosis that connects their hearing loss back to measles.
The AAO-HNSF clinical practice guideline for otitis media with effusion recommends watching and waiting for three months before referring a child for a hearing evaluation, while also recommending against screening for children who are not otherwise at risk.15 That is built-in delay that narrows the window for follow-up even in well-resourced settings. In an urban population that is insured, perhaps six of those nine children will eventually get to an audiologist. In the outbreak epicenter, that number drops to roughly three, because of the brick wall.3,4 Elzinga and colleagues’ systematic review found that the sensorineural component of OM-related hearing loss tends to concentrate at higher frequencies, where mild deficits are easily masked by concurrent conductive loss from the effusion itself, limiting detection even when a child sees an audiologist.14 Of the six urban children who make it to audiology, perhaps four will have the hearing loss documented. In the epicenter, that number is closer to two.
The last step in the funnel is where the numbers are the most striking, and where independent evidence from a completely different disease confirms the scale of the problem. When a child’s hearing loss is identified, the provider needs to know about that prior infection, think to connect it to the current presentation, and work in a system that supports retrospective linkage across time and providers. Wander and colleagues studied this exact problem in the context of long COVID, examining how often the VA health system successfully applied the ICD-10 sequela code U09.9 to patients with confirmed SARS-CoV-2 infections.12
The VA is the most integrated electronic health records system in the country, because the acute and follow-up records live in the same infrastructure. The answer was 5.28% at twelve months, which represents the best-case scenario for sequela coding, because you have a dedicated ICD-10 code created specifically for the purpose, a period of thorough clinical awareness, a shorter temporal gap between infection and sequelae than measles-to-hearing-loss typically involves, and a system where every provider can see every prior encounter.
Pfaff and colleagues found that even that 5% was not evenly spread out. The patients who received U09.9 codes skewed white, non-Hispanic, and toward low-poverty areas with high healthcare access, confirming that the same access gradients shaping the measles outbreak apply even when a purpose-built sequela code exists.13
Measles-associated hearing loss faces every barrier that long COVID sequela coding does, plus several more. As stated above, the timing between infection and complication can be months to years rather than weeks. Often it requires a specialty referral to audiology rather than primary care documentation. The coding workflow is more complex, built around B05.3 rather than a simpler add-on code. In an urban insured population, the model estimates that roughly 1 of every 1,000 measles cases will produce a hearing loss diagnosis linked back to measles in the coded record. In the outbreak epicenter, that number effectively rounds to zero.
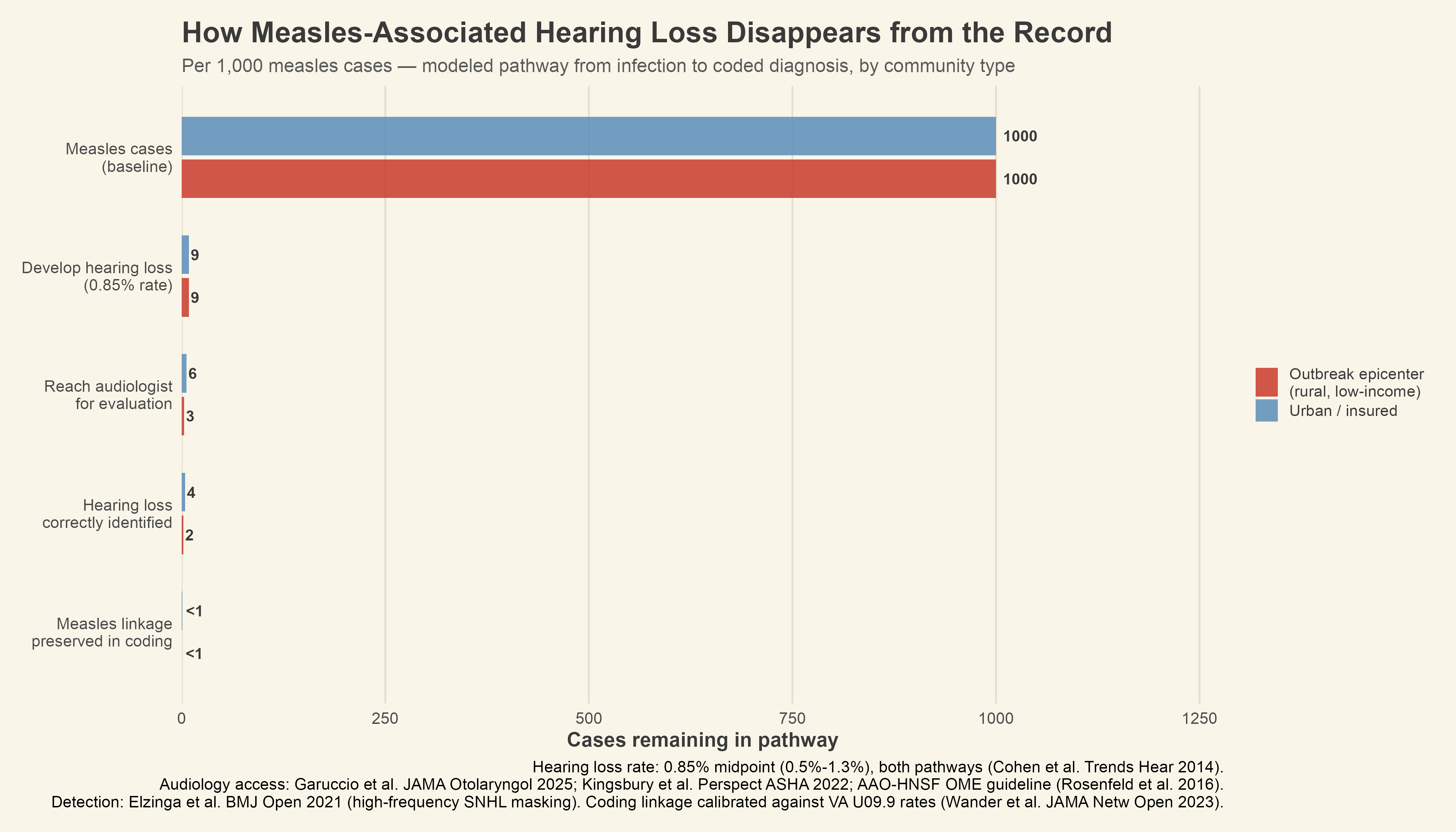
[Figure 4: Surveillance funnel, pathway from infection to coded measles-linked hearing loss diagnosis, urban vs. outbreak epicenter]
The HRSA Health Professional Shortage Area system scores counties from 0 to 25 on primary care access. Gaines County does not appear in the geographic HPSA designation file, and the reason is worth thinking about for a moment. Formal designation requires an application process, and many isolated communities lack the administrative infrastructure to complete it.10 Not being on the shortage list is not evidence of adequate care.
Using HRSA’s Area Health Resources Files, Gaines scores near 20 on that scale, while Lubbock sits around 5.11 That difference translates into a gap in surveillance capture that is wider than I initially expected. The capture model places the urban no-shortage probability at roughly 18%, the VA integrated-system benchmark at roughly 5%, and the maximum-shortage endpoint below 1%. This model is conceptual, and I’m upfront about that. The numbers look tidy, but they’re rough estimates. The real gaps in evidence are bigger than the chart suggests.
What the model does accomplish is making the access gradient visible as a continuous function rather than two disconnected estimates. Applied to the outbreak, the Gaines County cases are projected to have produced roughly 3 hearing loss cases, of which fewer than one will ever appear in a coded record linked to measles.
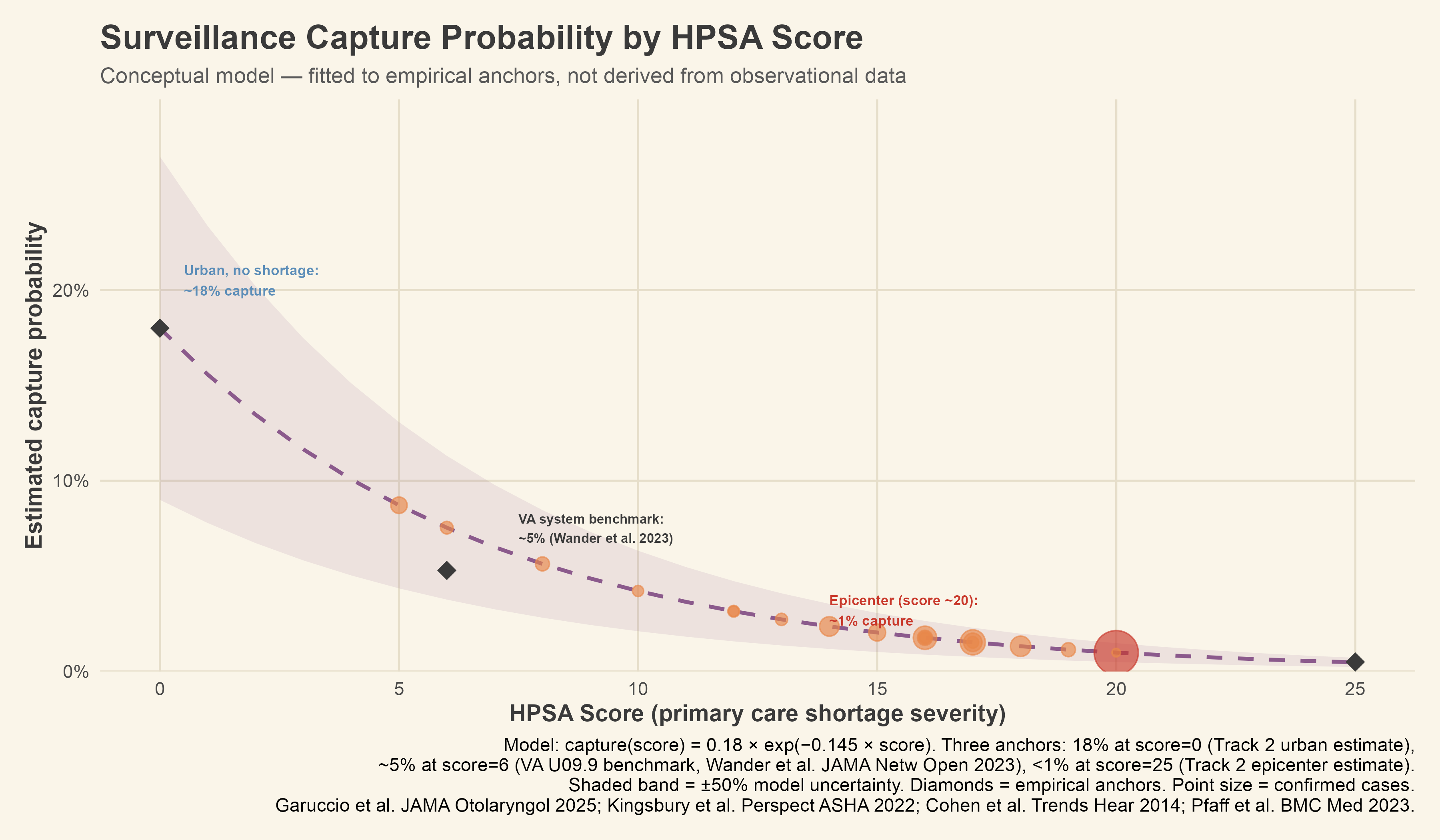
[Figure 5: Surveillance capture probability by HPSA score, with outbreak county estimates]
The long COVID comparison deserves a bit more exposure, because it gives us something that measles surveillance data cannot provide on its own, which is an independent benchmark. COVID-19 had every structural advantage that measles lacks when it comes to sequela documentation. The clinical community was aware of long-term complications from the earliest months of the pandemic. A dedicated ICD-10 code was created within two years. National screening programs were launched, and the VA system received billions of dollars in dedicated funding for post-COVID care.
Despite all of that, sequela coding captured roughly one in twenty confirmed cases. The 2025 measles outbreak had none of those advantages, played out in communities with fewer healthcare resources than the average VA facility, involved a complication pathway that takes months rather than weeks to manifest, and relied on a coding workflow that almost no provider in community practice has ever been trained to execute. The long COVID data does not prove that the measles capture rate matches the model. What it proves is that the model is not pessimistic. The true urban capture rate may sit closer to the VA’s 5% than to the estimated 18%, even in well-resourced settings.
The state-level picture provides context that is not easily brushed aside. Texas had an MMR kindergarten vaccination rate of 93.1% in the 2023-24 school year, below the 95% threshold associated with herd immunity. New Mexico came in at 94.0%, Oklahoma at 92.5%.5 All three outbreak cluster states fall below that threshold, and all three have substantial proportions of their counties designated as shortage areas. Vaccination gaps and healthcare access gaps tend to concentrate in the same communities for many of the same reasons, and an outbreak that starts where vaccination is low will tend to burn hottest in exactly the places least equipped to document what happens next.
Somewhere in Gaines County right now, there are children who had measles last year and whose families have not yet noticed that something is off. Maybe a child isn’t responding the way she should, or a teacher has said something, or a parent is carrying that low hum of concern. Some of those children will eventually get to an audiologist, and some will not. The ones who do will receive a hearing loss diagnosis coded under H90, and the measles will not be mentioned or not a contributing factor, because by then the acute encounter will be months or years in the past and the provider will have no reliable way of knowing exactly what happened. The cause will be listed as unknown.
What happened to me in 1977 played out again (and in South Carolina is still playing out) in a county with no audiologist, among children whose families did not vaccinate, inside a healthcare system that still has no reliable way to connect a hearing loss to the infection that caused it. The codes are right there in the book. The knowledge is all over the literature. The actual infrastructure to connect them in real life is missing.
Careful attention to data is what brings this to light. Not simply the fact that measles causes hearing loss, which is in every audiology textbook, but the specific location of the break in the causal chain between infection and documented outcome, and the way that break falls unevenly across geography and access. Consider the most basic question you might want to ask. Did hearing loss increase in the years following major measles outbreaks? The pre-vaccine era should have produced a detectable signal in population-level hearing loss data, if the coding infrastructure had ever captured it.
The 2019 Rockland County outbreak, the largest since elimination, should show up as a downstream cluster of pediatric hearing loss diagnoses in affected zip codes, if the follow-up visits were coded with any etiological linkage at all. The 2025 Texas data won’t show up fully in any claims database for another year or two, and when it does, the H90 codes will arrive stripped of context, with no measles encounter attached, indistinguishable from every other cause of childhood hearing loss. The question cannot be answered not because the data is incomplete, but because the system was never designed to answer it fully. That is not a limitation of this analysis, but a finding.
So what would it take to fix this?
The problem has three layers, and each one needs a different kind of intervention. The first is the coding workflow itself. Right now, sequela linkage depends on individual providers remembering that a prior infection occurred and knowing how to apply B05.3 or its equivalent in the correct clinical context. That is not a realistic expectation in a system where the acute encounter and the follow-up visit happen months apart, in different facilities, with different providers who may have no shared record. The fix is not education, but automation.
EHR systems already generate clinical decision support alerts for drug interactions and duplicate orders. There is no reason they cannot flag a patient with a documented measles encounter when that patient later presents with audiological symptoms, prompting the provider to consider sequela coding. The code exists. The workflow to trigger it does not, and building one is a software problem, not a medical one.
ICD-11, which the WHO released for global implementation in 2022, addresses part of this problem at the classification level. Its postcoordination feature allows coders to cluster multiple codes into a single expression that preserves causal relationships, so a hearing loss diagnosis could be formally linked to a prior measles infection within the coded record itself rather than relying on separate codes that happen to appear in the same chart.16 A generic sequela code, QC50, makes this possible for any prior condition causing current symptoms, which is a real structural improvement over ICD-10’s scattered precoordinated sequela codes.
The catch, and it is a doozy, is that the United States has not adopted ICD-11 and has no implementation date penciled in. The NCVHS workgroup studying the transition only began in 2023, no proposed rulemaking has been published, and the U.S. withdrawal from the WHO in January 2026 has caused additional uncertainty about whether and when adoption will happen.17 In context, the U.S. adopted ICD-10-CM in 2015, a full 26 years after the WHO finalized ICD-10 in 1989.
Even if ICD-11 arrives faster than that, the children in Gaines County will be adults before it does. More importantly, the authors of the ICD-11 sequelae coding literature are clear about the limitation that matters most here. As Drösler and colleagues wrote in August 2025, “those who generate the data need to know about this sequelae coding rule,” and “there is considerable potential for data errors, both in generation and analysis.”16 The codebook is getting better, but it has never been the problem.
The second layer is follow-up protocols. When a notifiable disease like measles is confirmed, public health agencies already conduct contact tracing and exposure investigation. What they do not do, in any jurisdiction I have been able to identify, is trigger downstream screening for known complications.
A confirmed pediatric measles case should generate an automatic audiological referral at three and six months, the same way a confirmed bacterial meningitis case triggers hearing screening before hospital discharge.18 The clinical basis for this is well established. The operational mechanism for doing it at scale is not, because outbreak response and longitudinal follow-up live in different administrative silos. Bridging that gap would require state health departments to build complication-tracking workflows into their case investigation systems, which would cost money and attention, both of which are in short supply during an active outbreak.
The third layer is the one that is hardest to fix because it involves the communities where these outbreaks concentrate. Gaines County has no audiologist. The children most likely to develop measles-associated hearing loss are the same children least likely to reach the specialist who will keep the causal link alive. Garuccio and colleagues’ finding that 75% of U.S. counties are hearing healthcare shortage areas means this is not a problem that can be solved by telling families to drive to Lubbock.3
Teleaudiology has expanded since the pandemic, and mobile screening programs have shown promise in school-based settings, but neither has been deployed at the scale or with the targeting that would make a difference in the aftermath of a rural outbreak. The infrastructure gap is real, and closing it means investment that goes well beyond what a coding fix or a protocol change can accomplish.
None of these interventions are exotic, because they are all technically feasible with existing technology and clinical knowledge. What they lack is the political and institutional momentum that comes from being able to point to a number and say, this is how many children were harmed, and this is how we know.
The entire point of this article is that the number does not exist, because the system that would produce it was never built. If we want to have the number for the next outbreak, we have to build the system now, while the memory of this one is still fresh and the children in Gaines County are still young enough for early intervention to make a difference.
That gap between infection and record is not an abstraction for me. The answer to my own hearing loss was sitting in the epidemiology of 1977 the whole time, buried under a missing rash and a fever my mom still can't forget.
References
- Cohen BE, Durstenfeld A, Roehm PC. Viral causes of hearing loss: a review for hearing health professionals. Trends Hear. 2014;18:2331216514541361. doi:10.1177/2331216514541361
- Mathis AD, Raines K, Filardo TD, et al. Measles update — United States, January 1–April 17, 2025. MMWR Morb Mortal Wkly Rep. 2025;74(14):232-238. doi:10.15585/mmwr.mm7414a1
- Garuccio J, Ukert B, Arnold M, Phillips S, Pesko MF. Using supply and demand to identify shortages in the hearing health care professional workforce. JAMA Otolaryngol Head Neck Surg. 2025;151(9):868-873. doi:10.1001/jamaoto.2025.2112
- Kingsbury S, Khvalabov N, Stirn J, et al. Barriers to equity in pediatric hearing health care: a review of the evidence. Perspect ASHA Spec Interest Groups. 2022;7(4):1060-1071. doi:10.1044/2021_PERSP-21-00188
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated 2025. https://www.cdc.gov/measles/cases-outbreaks.html. Accessed February 19, 2026.
- Centers for Disease Control and Prevention. Measles data and statistics. https://www.cdc.gov/measles/data-research/index.html. Accessed February 27, 2026.
- Centers for Disease Control and Prevention. Summary of notifiable diseases, United States, 1993. MMWR Morb Mortal Wkly Rep. 1993;42(53).
- Texas Department of State Health Services. Measles Outbreak Investigation: Final Report. Texas DSHS; 2025.
- Pan American Health Organization/World Health Organization. Disease outbreak news: measles — United States of America. Published April 17, 2025. https://www.paho.org/en/documents/disease-outbreak-news-measles-united-states-america-17-april-2025. Accessed February 25, 2026.
- Health Resources and Services Administration. Bureau of Health Workforce: health professional shortage areas. https://data.hrsa.gov/topics/health-workforce/shortage-areas. Accessed February 20, 2026.
- Health Resources and Services Administration. Area Health Resources Files, 2022-2023. HRSA Bureau of Health Workforce; 2023. https://data.hrsa.gov/topics/health-workforce/ahrf. Accessed February 16, 2026.
- Wander PL, Baraff A, Fox A, et al. Rates of ICD-10 code U09.9 documentation and clinical characteristics of VA patients with post-COVID-19 condition. JAMA Netw Open. 2023;6(12):e2346783. doi:10.1001/jamanetworkopen.2023.46783
- Pfaff ER, Madlock-Brown C, Baratta JM, et al. Coding long COVID: characterizing a new disease through an ICD-10 lens. BMC Med. 2023;21(1):58. doi:10.1186/s12916-023-02737-6
- Elzinga HBE, van Oorschot HD, Stegeman I, Smit AL. Relation between otitis media and sensorineural hearing loss: a systematic review. BMJ Open. 2021;11(8):e050108. doi:10.1136/bmjopen-2021-050108
- Rosenfeld RM, Shin JJ, Schwartz SR, et al. Clinical practice guideline: otitis media with effusion (update). Otolaryngol Head Neck Surg. 2016;154(1 Suppl):S1-S41. doi:10.1177/0194599815623467
- Drösler SE, Weber S, Chute CG. New ICD-11 features for coding late sequelae and chronic post-procedural conditions. BMC Med Inform Decis Mak. 2025;25(1):318. doi:10.1186/s12911-025-03121-5
- National Committee on Vital and Health Statistics. Workgroup on Timely and Strategic Action to Inform ICD-11 Policy. https://ncvhs.hhs.gov/subcommittees/workgroup-on-timely-and-strategic-action-to-inform-icd-11-policy/. Accessed February 15, 2026.
- Joint Committee on Infant Hearing. Year 2019 position statement: principles and guidelines for early hearing detection and intervention programs. J Early Hear Detect Interv. 2019;4(2):1-44. doi:10.15142/fptk-b748
A note on methods: All hearing loss projections in this piece use the 0.5% to 1.3% complication rate from Cohen et al. (2014), which encompasses both the otitis media pathway (middle ear infection progressing to sensorineural damage) and the direct cochlear damage pathway (viral destruction of hair cells and spiral ganglion neurons). These are applied to CDC case and true-infection estimates to produce modeled projections, not measured outcomes. The surveillance funnel model (Figure 4) was calibrated using long COVID sequela coding rates from the VA health system (Wander et al. 2023) as an independent benchmark for the coding linkage step. The capture probability curve (Figure 5) is a conceptual model fitted to three empirical anchors rather than derived from observational surveillance data; it is presented with a wide uncertainty band to reflect that distinction. The author’s personal medical history is included as context and framing, not as evidence for any empirical claim.
Code and additional data sources available at https://github.com/NotesfromtheAbstract/measles-hearing-loss-surveillance.

